Neuromodulation of sensory circuits
Our group is interested in how neuromodulation reshapes sensory circuits to make them more adaptable and better suited for a specific environment. Neuromodulators are chemicals produced by neurons in the brain and released in either a synapses specific manner or more broadly as a diffusible neurohormone. These chemicals can change the intrinsic properties of individual neurons in a circuit and thus alter how the neural circuit functions. Such perturbations of network functioning lends flexibility to the nervous system and allows to function of a wide range of conditions. However, several neuropathologies in humans are suspected to have causes attributed to the dysfunction of modulatory systems; such as serotonin's role in anxiety and depression and dopamine's role in Parkinson's disease and Schizophrenia.
We use the model system Drosophila to investigate the question of how neuromodulators rewire the brain to better perform certain tasks. Specifically, we are focusing on the role of various modulators in the sense of smell, known as olfaction. The Drosophila olfactory system is the ideal system to tackle these challenges due to 1) the availability of a broad genetic toolkit to perturb genes, 2) the reduced number of neurons in the fly olfactory system, 3) and finally its highly analogous organization compared to mammalian olfactory systems.
We use the model system Drosophila to investigate the question of how neuromodulators rewire the brain to better perform certain tasks. Specifically, we are focusing on the role of various modulators in the sense of smell, known as olfaction. The Drosophila olfactory system is the ideal system to tackle these challenges due to 1) the availability of a broad genetic toolkit to perturb genes, 2) the reduced number of neurons in the fly olfactory system, 3) and finally its highly analogous organization compared to mammalian olfactory systems.
Tools and Techniques
|
Whole-cell Physiology - The main tool employed in the lab to dissect brain circuitry in the fly is whole-cell patch-clamp electrophysiology. This technique allows us to record the electrical activity of individual neurons in the fly nervous system. We can target specific neurons in the brain fro recording by expressing green fluorescent protein exclusively in cells of interest. These recordings are generally obtained from intact preparations thus allowing us to correlate cellular activity with sensory stimulation. Whole-cell patch clamping is considered the gold standard for measuring the electrical activity of neurons.
|
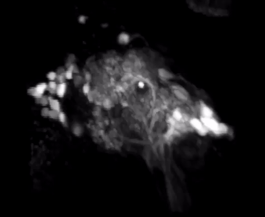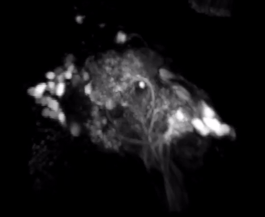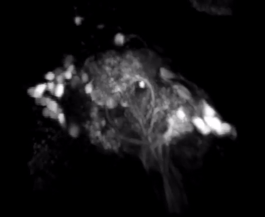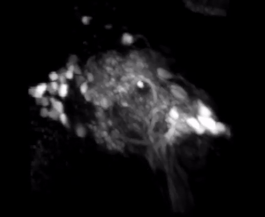
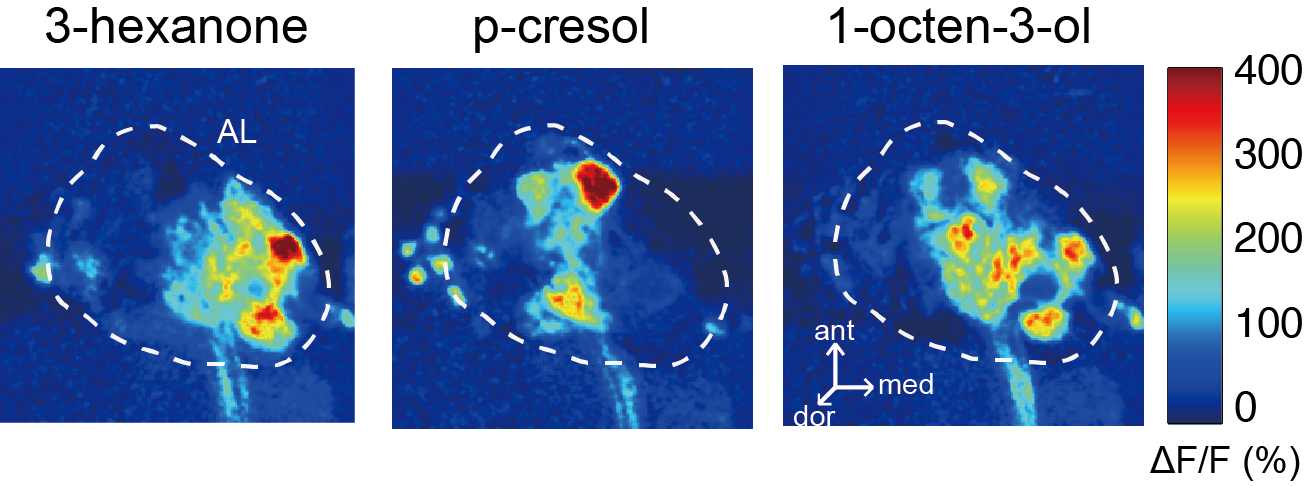
2-Photon Microscopy - We use 2-photon microscopy for a variety of experiments in the lab. For example, this powerful tool allows to record the activity of large populations of neurons as measured via calcium indicators such as GCaMPs. Alternatively, we can measure the spatial distribution of calcium signals within the dendritic arbors of individual neurons. This resolution is not easily achieved using conventional electrophysiological approaches. Finally, we can also use 2-photon laser microscopy for anatomical tract-tracing with photoactivatable GFP or for ablating specific axon tracts to test their functional significance.
|
Quantitative Animal Behavior - We are developing quantitative tools to precisely link our physiological experiments with sensory perceptions, as measured through behavior. We utilize two separate approaches to characterize behavior in the lab. The first relies on using image processing algorithms to precisely annotate the behavior or freely roaming animals. This approach is suitable to high-throughput screen. It's main limitation is the difficulty in controlling the stimuli that the animal encounters. We have thus developed a second behavioral paradigm where the animal is positioned on a small sphere suspended on a cushion of air while we deliver it olfactory stimuli in a highly precise fashion. The animals locomotory intentions can be inferred by using optical sensory to capture the movements generated in the sphere.
|
We are extremely grateful for the generous funding that we have received to from the NIH NIDCD and Whitehall Foundation that supports our work